The "Off" Button That Lets Plants Make Flowers
Research led Doris Wagner, Robert I. Williams Term Professor of Biology, identified a gene that, when stifled, generates blooms.

While most animals are born with all the organs they’ll ever have, plants generate new organs, including flowers, leaves, and branches, throughout their lifespan. A new study of flower formation by Doris Wagner and her lab shows how one molecular pathway is stifled in order for blooms to arise—a key step in plant reproduction.
“Identity is not just what you are; it’s what you aren’t,” says Wagner, Robert I. Williams Term Professor of Biology. “There are so many alternative fates that any cell or tissue type could have in an organism that aren’t realized, and that they’re not realized is part of that identity.”
In animals, it’s been known that pathways governing stem cells must be repressed in order to give rise to organs. The new findings, published in the journal Nature Communications, represent the first time that the same phenomenon has been demonstrated in plants.
Wagner’s lab has long focused on flower formation, given its central role in both reproduction and agricultural yield. Recently, they’ve turned attention to a group of enzymes known as auxin response factors (ARFs), which regulate the transcription of DNA into RNA and respond to auxin, a plant hormone that governs a wide range of aspects of plant development. One ARF is MONOPTEROS (MP), known to activate genes that promote flower formation.
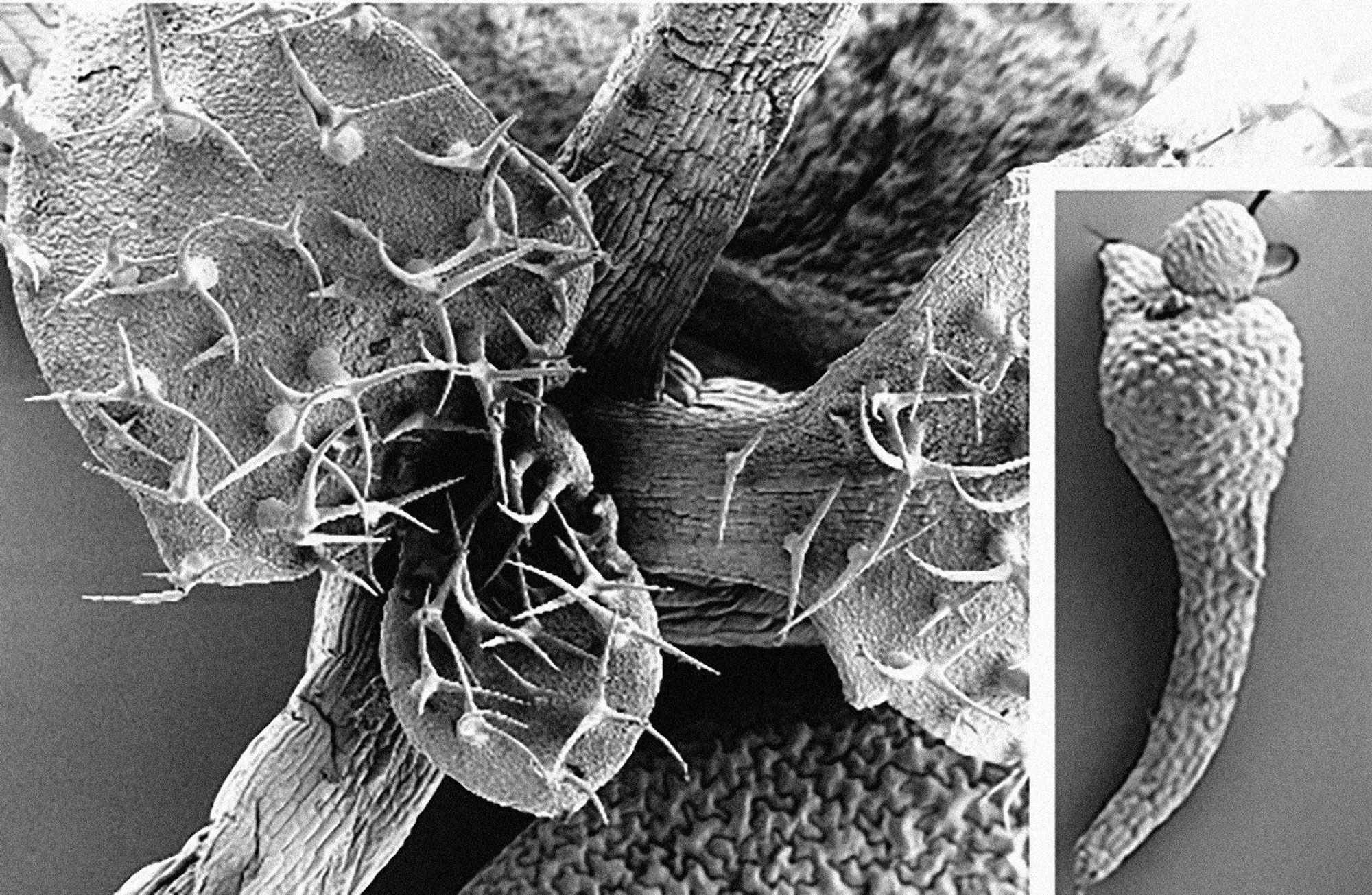
Guided by research from other groups, Wagner’s team studied two other ARFs that had been found to repress developmental pathways in plants, ETT and ARF4. They noticed that these genes were expressed in the primordial “founder” cells of the plant—those that give rise to flowers—suggesting the ARFs may play a role in flower formation. When Wagner and colleagues created a mutant plant that lacked normal levels of MP, ETT, and ARF4, they wound up with very odd, triangle-shaped plants that failed to develop any flowers, leaves, or branches.
“We got this very severe phenotype where the plants don’t make any lateral organs from the embryo,” says Wagner, who coauthored the paper with colleagues from Penn, Norwich Research Park; and Centro de Investigación y de Estudios Avanzados del Instituto Politécnico Nacional. “You have this very large meristem, the pocket where stem cells are harbored. Clearly stem-cell fate, or pluripotency, needs to be shut off in order for organogenesis to occur.”
The team also observed that levels of SHOOT MERISTEMLESS (STM), a gene involved in maintaining a pool of stem cells, which can give rise to a variety of cell types, was highest in the triple mutant. To test whether STM blocked organ development, the researchers artificially enhanced levels of STM. The resulting plants made even more flower-less “pin” stems; in contrast, blocking STM allowed flowers to form.
Further probing revealed that MP acted indirectly to repress STM, working through an intermediary gene, FIL. ETT and ARF4, meanwhile, acted directly to repress STM and give rise to flowers. ARF4, ETT, and FIL, the researchers found, were each able shut off STM by recruiting enzymes that compress chromatin—tightly packed DNA—to the gene, preventing it from being expressed.
Wagner and her team are still uncovering the details of this process and considering its implications. She notes that STM is involved in influencing the shape of leaves, which are simpler in form when STM is shut off. This species-specific characteristic is used for plant classification, gives rise to a plethora of different ornamental plant forms, and underscores the centrality of the STM “off button,” a ripe topic for further research.
The study was supported in part by the National Science Foundation (Grant 1557529).